Product Regulatory Compliance: Issues & Needs 2 – Expirations and Events
By Tom Killam and Cyril Mecwan
July 9, 2021 | Series 1 / No. 6
The ongoing recertification of expiring regulatory certificates can become a mini operation, requiring crisp management of renewal projects, resources, money and possibly test samples. Having an expiration of a Type Approval can result into ‘Stop ship’ or ‘Ship hold’ if not managed properly.
The product regulatory compliance processes and events take place throughout the lifecycle of a product. If not managed on a day-to-day basis, these seemingly mundane events bring disarray to the overall organization and functioning of the space.
The Event Based Management automates the notifications of these expirations and events, prompts the users to take actions, and guides them through the decision-making process. A product regulatory compliance professional stands to gain big by the use of Event Based Management.
Background:
Keeping track of the upcoming expirations for the purpose of planning, budgeting, and obtaining the recertification before the expiration, is a significant effort and can become a problem if not processed prior to expiration.
When an OEM is selling a product to multiple countries, many projects, transactions, and events take place at a large scale. And when the OEM is releasing many products in many markets, one is dealing with an even larger scale. These seemingly mundane events bring disarray to the overall organization and functioning of the space if not managed on a day-to-day basis.
Issues and Needs:
Expirations: Some regulatory authorities grant a product regulatory certificate for the life of the product (Japan – VCCI), whereas some grant with a validity date for a year (Mexico – NOM), two years (Brazil - ANATEL), three years (S. Africa - NRCS), or five years (India - BIS). Underwriter’s Laboratory (UL) issues a regulatory certificate for life, however, it remains valid as long as the subsequent factory inspections performed by the UL Inspector are satisfactory.
It is critically important that the expiring certificates be renewed in time, otherwise it may result into ‘Stop ship’ of the product, or even worst ‘Ship hold’ at the customs of the incoming country. This in turn has a negative impact on revenue and customer satisfaction.
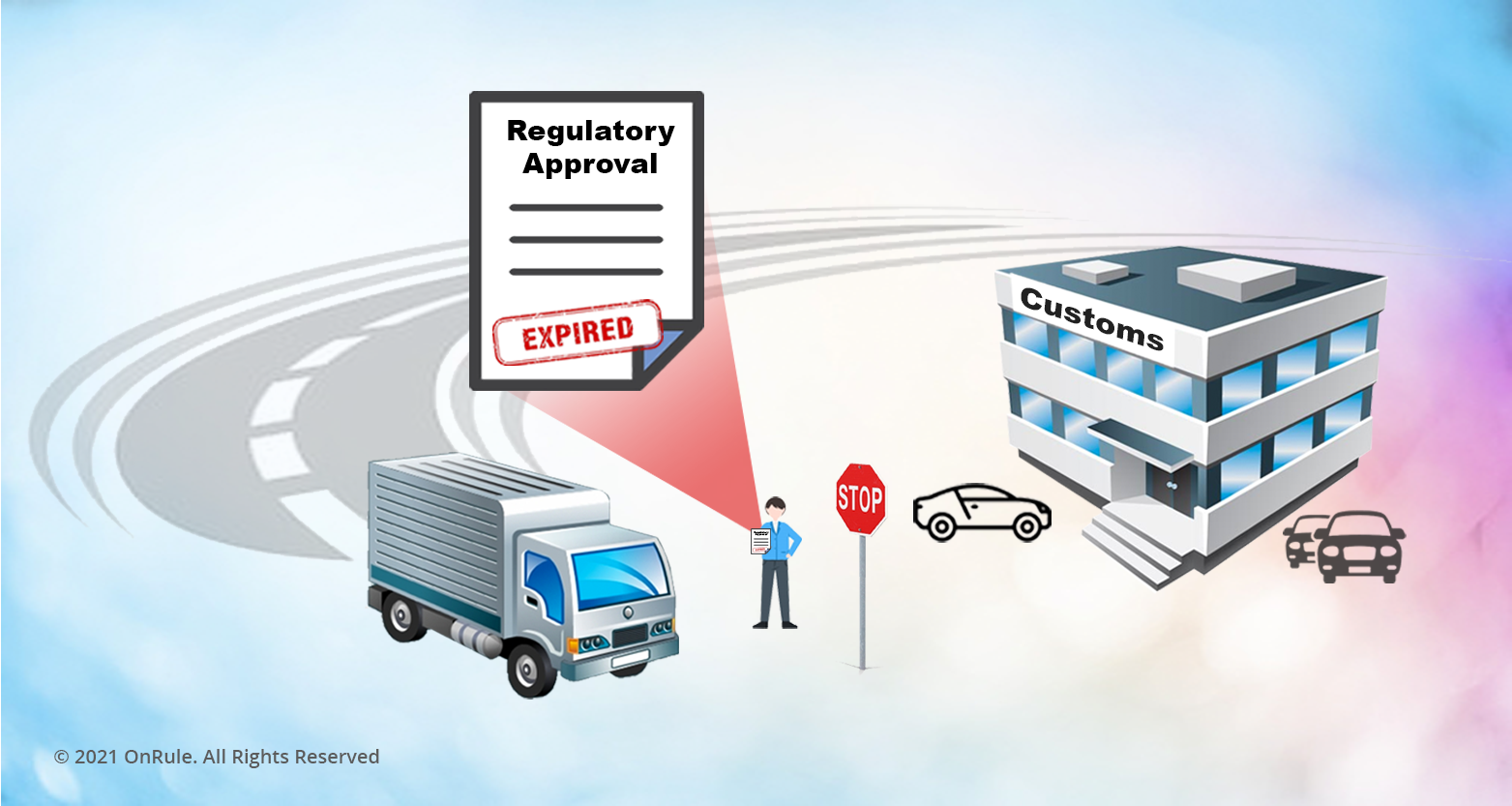
The mid-size to large companies selling multiple products into many markets may experience multiple expirations on different products every month. The ongoing recertification task can become a mini operation, requiring crisp management of renewal projects, resources, and money. Because there is a lead time associated with the testing of the product for the purpose of recertification, a proactive management requires that the renewal is completed well before the expiration, avoiding any last-moment operational risk.
Therefore, for the purpose of better planning a recertification activity, an alert mechanism or a report outlining upcoming and month-wise expirations is desired that may aid in overall planning of resources and allocation of budget.
Events: Obtaining and managing product regulatory compliance is a highly collaborative activity. A Compliance professional interacts with the Engineering, New Product Introduction Engineer (NPIE), Product Management, Distributors or Importers, various test lab personnel, etc. during the process of managing a new product certification or recertification.
A Compliance professional would like to receive automated messages informing him/her about the progress made on the project, and notifications for any impending actions. For instance, when a responsible/ authorized person signs a self-declaration (DoC), the Compliance professional would like to be notified opposed to personally chasing the signatory or passively awaiting the signature. Similarly, if a test report is shared by the test lab for the review, or if a question is asked by a peer on a particular project, the Compliance professional would like to be automatically informed of these actions or query. In yet another example, if a standard that is used by the OEM is changing, the Compliance professional would like to receive an intelligent notification that informs him/her of the upcoming change as well as a possible impact to his/her product and records portfolio.